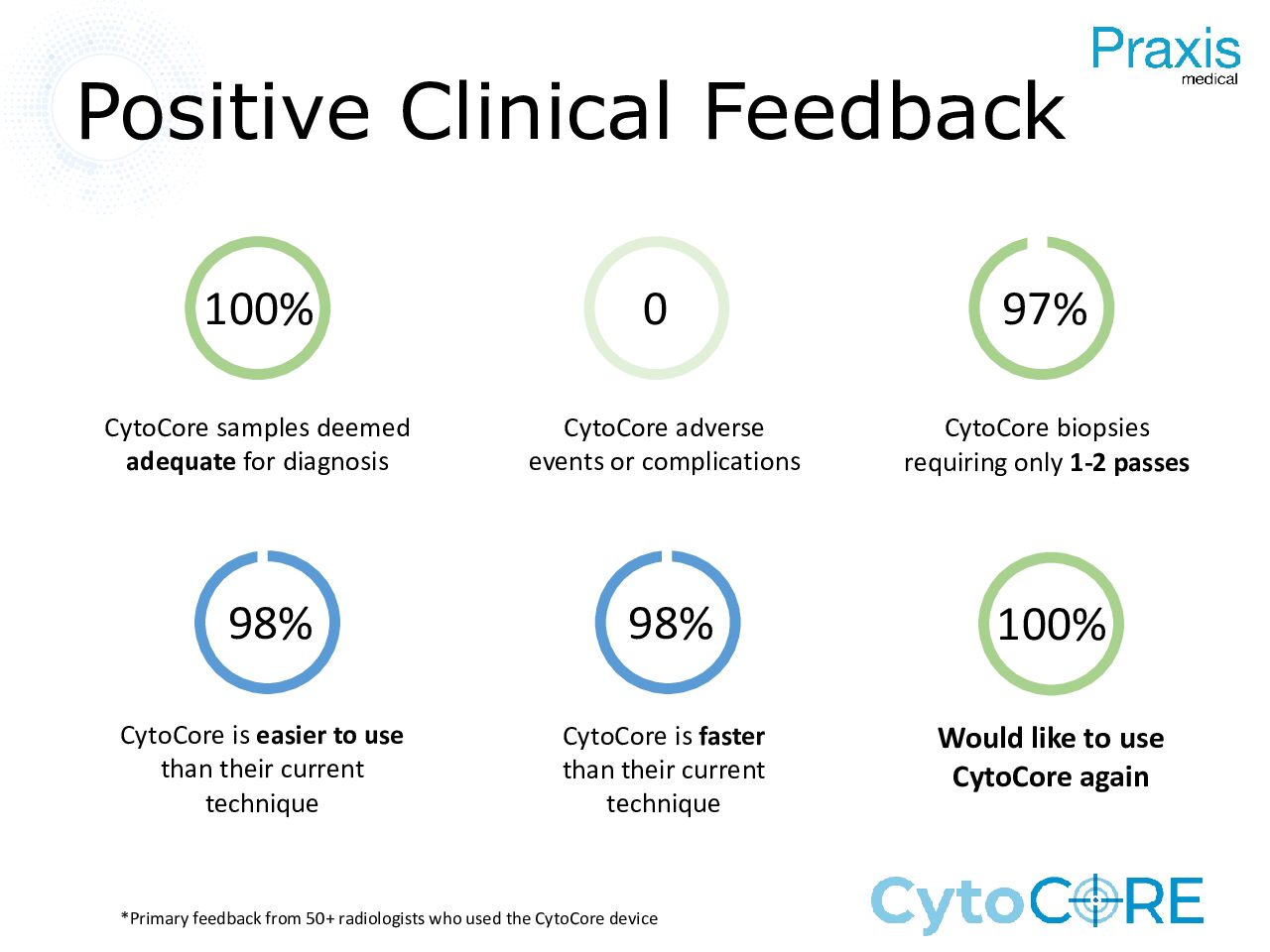
Clinical Data
Motorized Rotating Fine Needle Biopsy Device Reduces Number Passes Needed for Cytological Adequacy and Improves Diagnostic Accuracy
Summary
This study aimed to determine the impact of using CytoCore® on the relative number of passes required to obtain an adequate sample and a diagnostic biopsy in comparison to global averages using traditional FNA. Results showed that CytoCore® improves clinical efficiency by reducing the number of passes required to obtain an adequate sample and reducing the rate of nondiagnostic diagnoses.
Key points
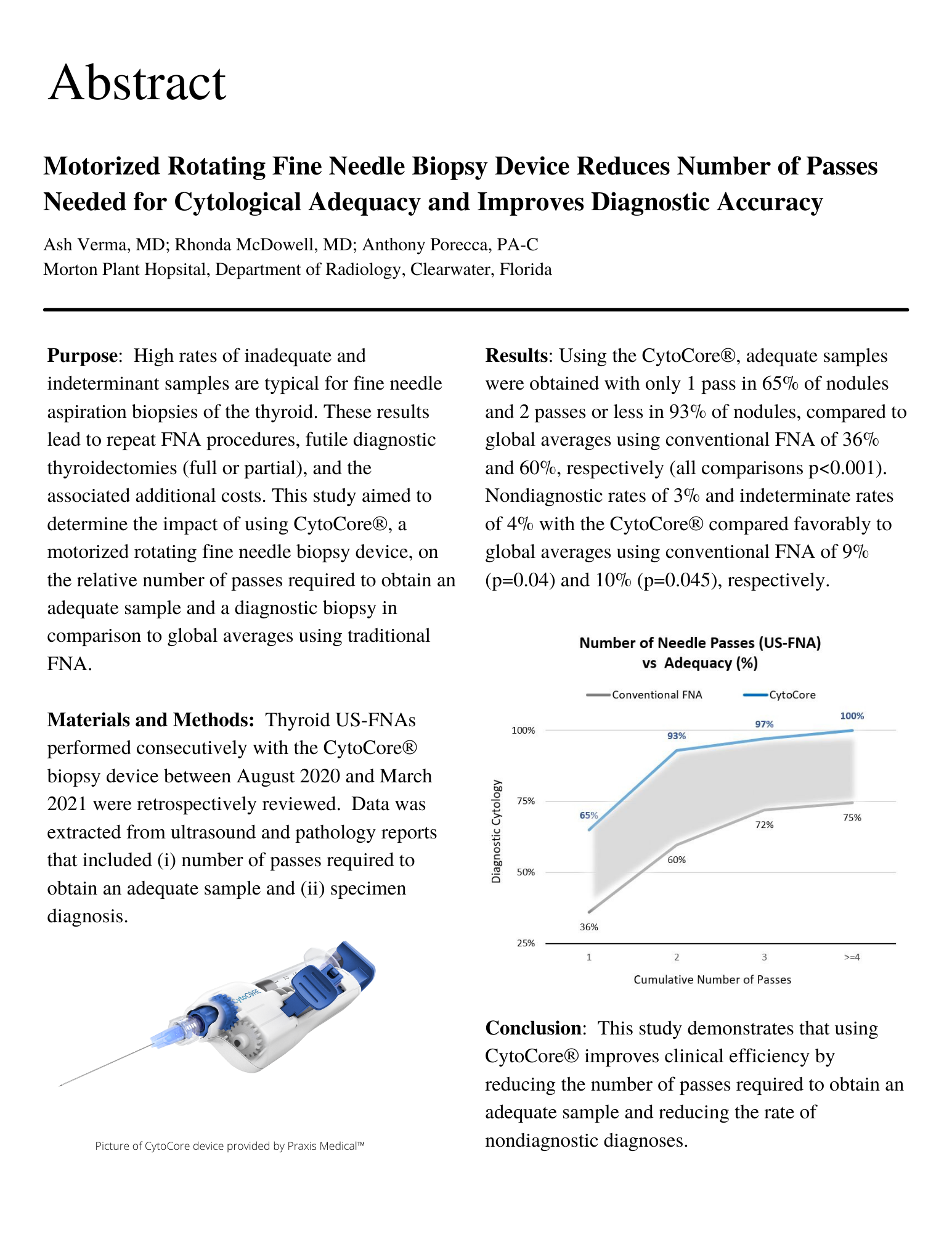
Using the CytoCore®, adequate samples were obtained with only 1 pass in 65% of nodules and 2 passes or less in 93% of nodules, compared to global averages using conventional FNA of 36% and 60%, respectively (all comparisons p<0.001).
Diagnostic rates of 3% and indeterminate rates of 4% with the CytoCore® compared favorably to global averages using conventional FNA of 9% (p-0.04) and 10% (p-0.045), respectively.
Contact us for a copy of the study or for more information.
Increasing the Cellular Yield of Pathology Samples Through a Motorized Rotating, Aspirating Device
Summary
Key points
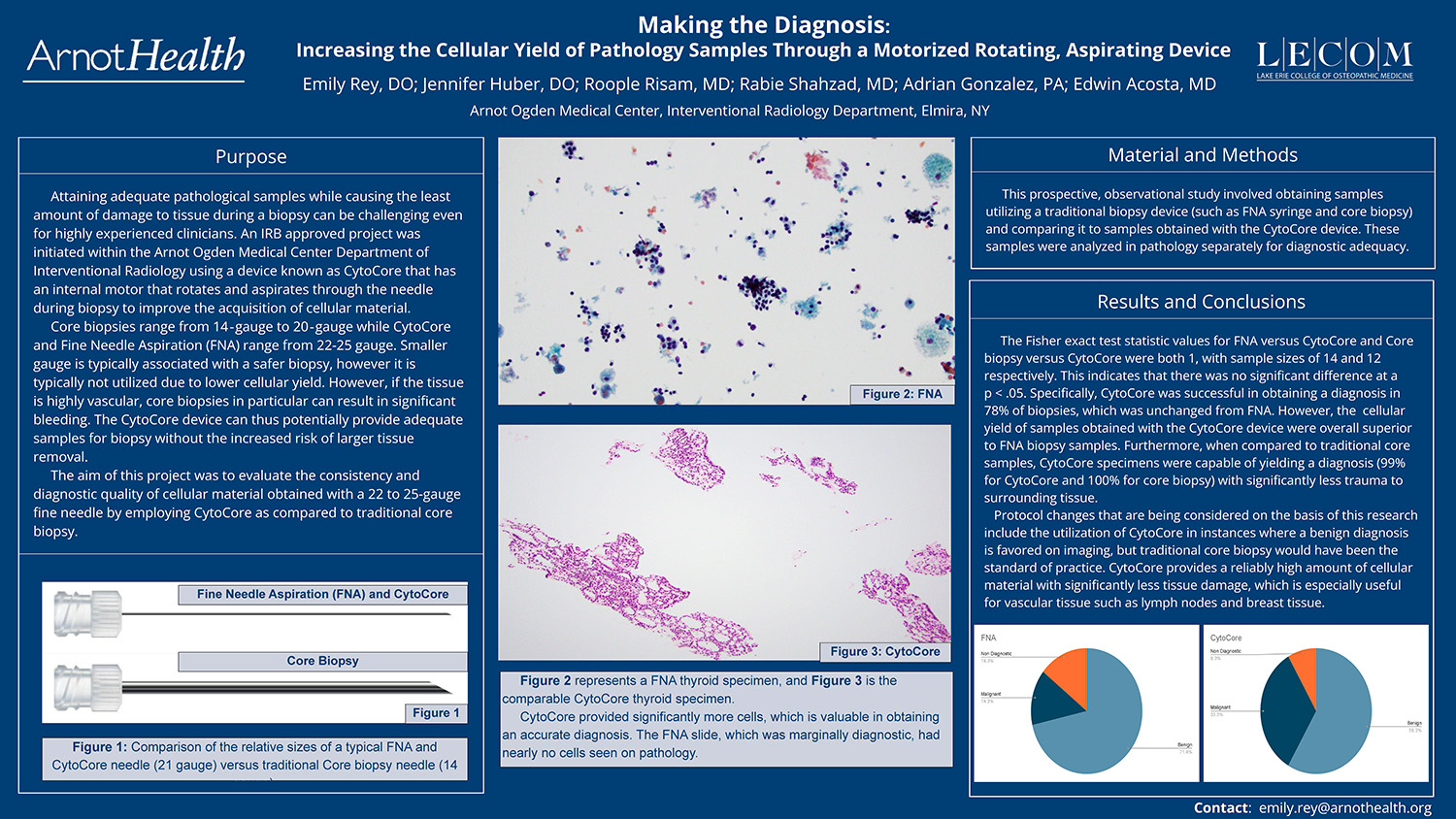
The cellular yield of samples obtained with the CytoCore device were overall superior to FNA biopsy samples.
When compared with traditional core biopsy samples, CytoCore specimens were capable of yielding a diagnosis (99% for CytoCore and 100% for core biopsy) with significantly less trauma to surrounding tissue.
Contact us for a copy of the study or for more information.
White Paper Summary: CytoCore USF Live Pig Study
Summary
The purpose of this study was to
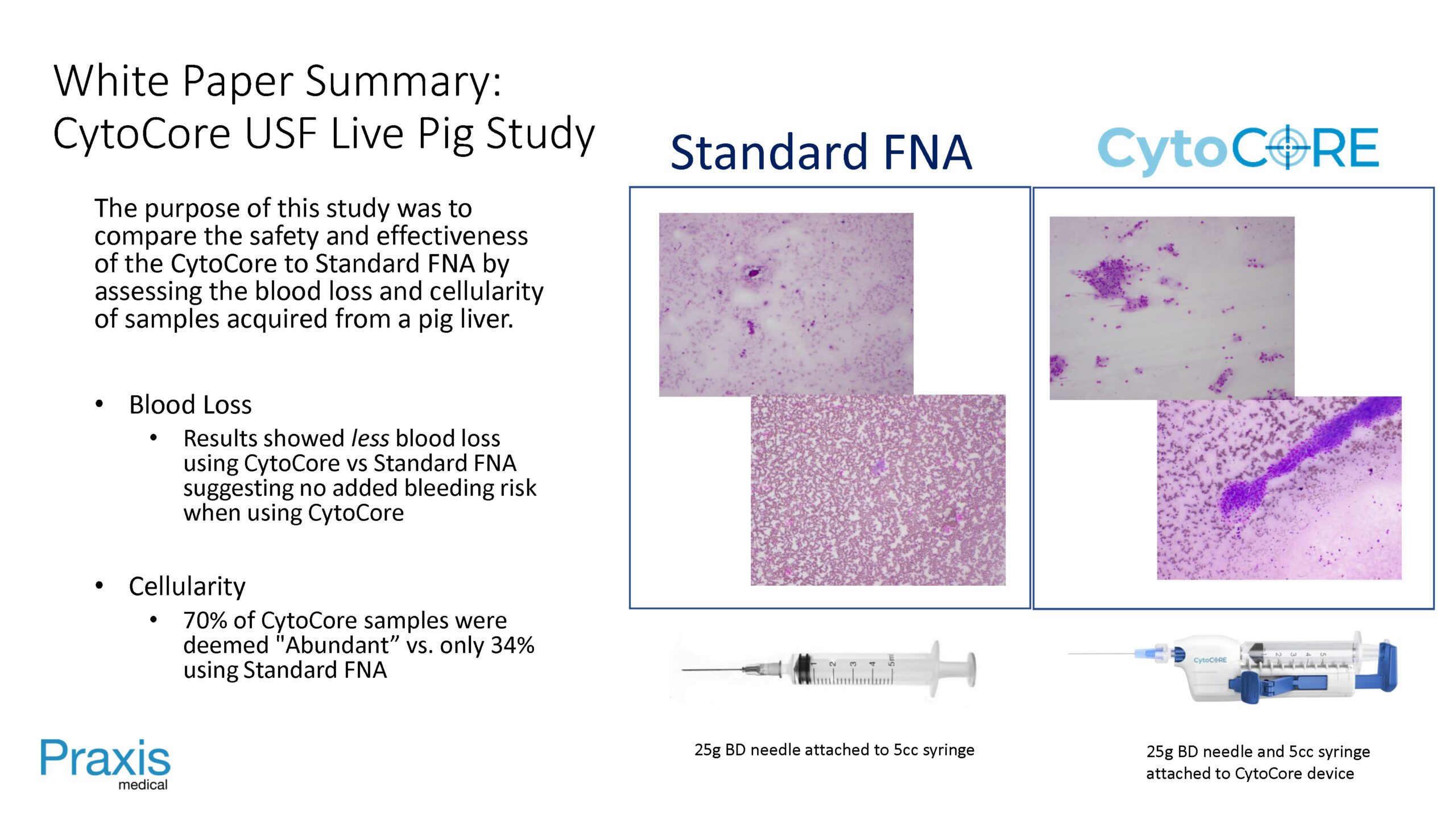
compare the safety and effectiveness of the CytoCore to Standard FNA by assessing the blood loss and cellularity of samples acquired from a pig liver.
Key points
Blood Loss: Results showed less blood lossusing CytoCore vs Standard FNA suggesting no added bleeding risk when using CytoCore
Cellularity: 70% of CytoCore samples were deemed “Abundant” vs. only 34% using Standard FNA
Contact us for a copy of the study or for more information.
CytoCore Lung Biopsy Case Study
Summary
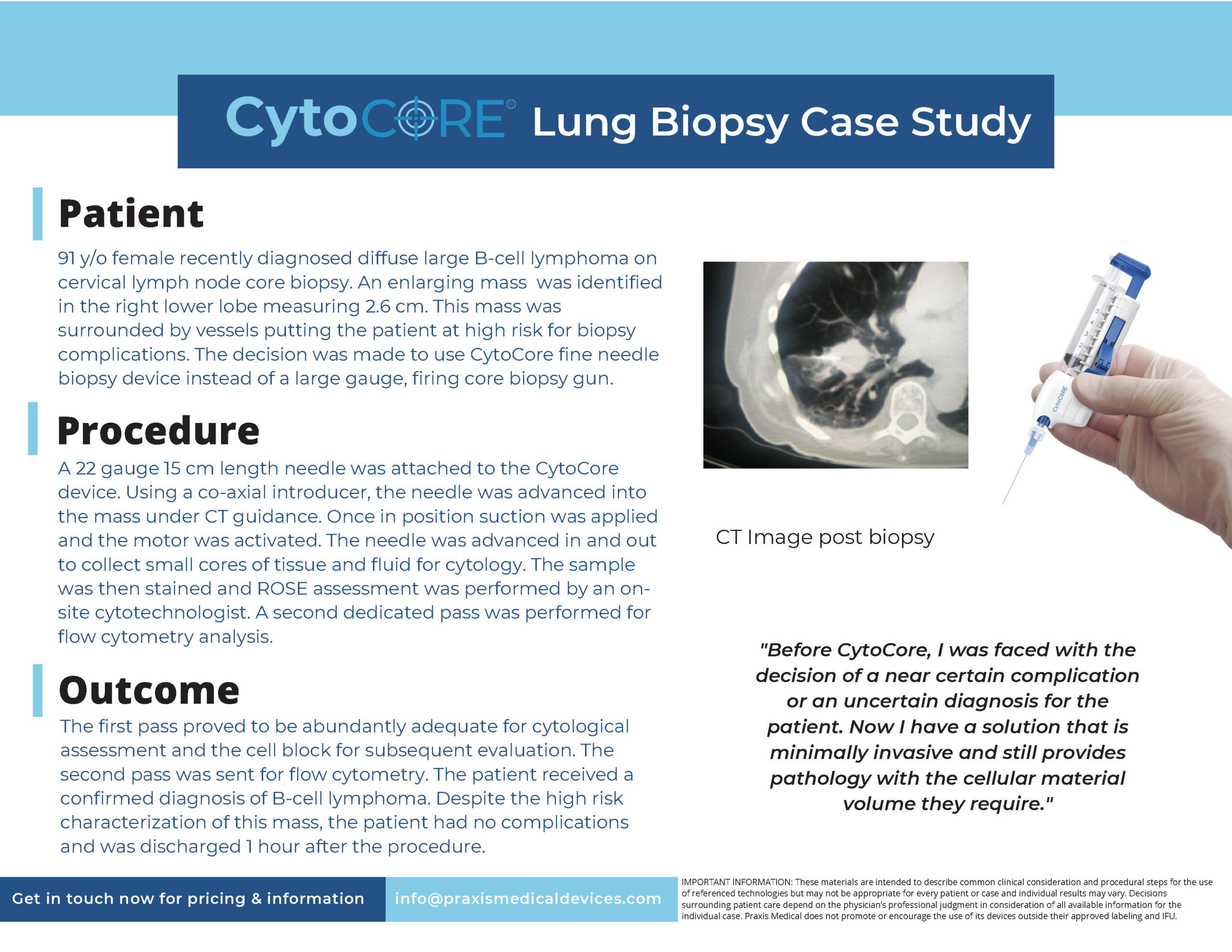
91 y/o female recently diagnosed diffuse large B-cell lymphoma oncervical lymph node core biopsy. An enlarging mass was identifiedin the right lower lobe measuring 2.6 cm. This mass wassurrounded by vessels putting the patient at high risk for biopsycomplications. The decision was made to use CytoCore fine needlebiopsy device instead of a large gauge, firing core biopsy gun.
Key points
The first pass proved to be abundantly adequate for cytologicalassessment and the cell block for subsequent evaluation. Thesecond pass was sent for flow cytometry. The patient received aconfirmed diagnosis of B-cell lymphoma. Despite the high riskcharacterization of this mass, the patient had no complicationsand was discharged 1 hour after the procedure.
Contact us for a copy of the study or for more information.